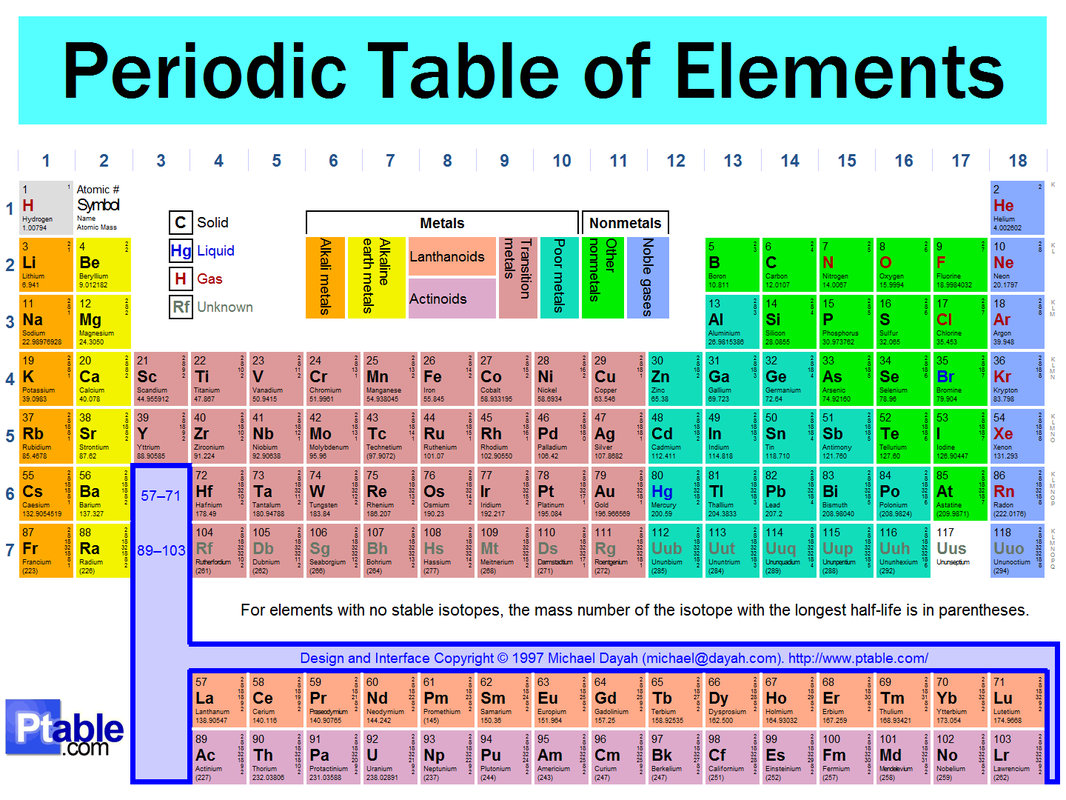
Groups, or families, in other words are the different columns and tell us how many valence electrons each group of elements have-
Periods are the rows of elements going from left to right. Groups can tell us exactly how many energy levels a specific element has.
Elements are classified by the following:
Valence Electrons: The elements are arranged in Groups, or families, which tell us how many valence electrons an element has.
Metals: Metals can be found in the middle of the periodic table of elements starting with group 3 and ending at group 16. But remember, there is a stair-step pattern starting with Aluminum and goes diagonally down from there.
Non-metals: Non-metals can be found in the upper-right-hand corner of the periodic table of Elements.
Metalloids: These can be found by looking at the stair-step pattern. they are the ones that touch the fine line.
Atomic #: Elements all have specific traits and one of those is the atomic number of the element. this tells us how many protons the atom has. And if it is neutral, it also tells us how many neutrons it has. Elements are arranged chronologically by this number.
Atomic Mass: The atomic mass of an atom is the protons + the neutrons. you can find this right under the symbol of an element.
You can figure out if the element that you are trying to find is reactive or not by this:
The reactive elements are located near the left and right sides. the least reactive ones are in the middle.
The properties of different types of elements are listed below:
Metals: these are classified by their hardness, which is very hard and almost solid.
Metalloids: these are in between metals and non-metals. they are also semiconductors.
Non-metals:
Elements are classified by the following:
Valence Electrons: The elements are arranged in Groups, or families, which tell us how many valence electrons an element has.
Metals: Metals can be found in the middle of the periodic table of elements starting with group 3 and ending at group 16. But remember, there is a stair-step pattern starting with Aluminum and goes diagonally down from there.
Non-metals: Non-metals can be found in the upper-right-hand corner of the periodic table of Elements.
Metalloids: These can be found by looking at the stair-step pattern. they are the ones that touch the fine line.
Atomic #: Elements all have specific traits and one of those is the atomic number of the element. this tells us how many protons the atom has. And if it is neutral, it also tells us how many neutrons it has. Elements are arranged chronologically by this number.
Atomic Mass: The atomic mass of an atom is the protons + the neutrons. you can find this right under the symbol of an element.
You can figure out if the element that you are trying to find is reactive or not by this:
The reactive elements are located near the left and right sides. the least reactive ones are in the middle.
The properties of different types of elements are listed below:
Metals: these are classified by their hardness, which is very hard and almost solid.
Metalloids: these are in between metals and non-metals. they are also semiconductors.
Non-metals: